Of all the terms that come up in conversations about GERD, Barrett’s esophagus is probably the one that causes the most anxiety. People hear it and immediately think of cancer. They worry that their years of heartburn have quietly been doing something irreversible. They search for it online at midnight and come away more frightened than informed.
That fear is understandable. But it is also, for the vast majority of people, significantly out of proportion to the actual risk.
Barrett’s esophagus is a real condition that deserves serious attention. It does increase the risk of a specific type of esophageal cancer. But the absolute risk of that progression for any individual with Barrett’s esophagus is much lower than most people assume, and with proper monitoring, the condition is highly manageable. The goal of this article is to give you an accurate, clear picture of what Barrett’s esophagus actually is, how it develops, who should be screened for it, and what happens if it is found.
This article is part of our GERD series. For the complete guide to acid reflux and GERD, visit our GERD and Acid Reflux Explained guide.
What Barrett’s Esophagus Actually Is
To understand Barrett’s esophagus, you need to understand what the normal esophageal lining looks like and why it changes.
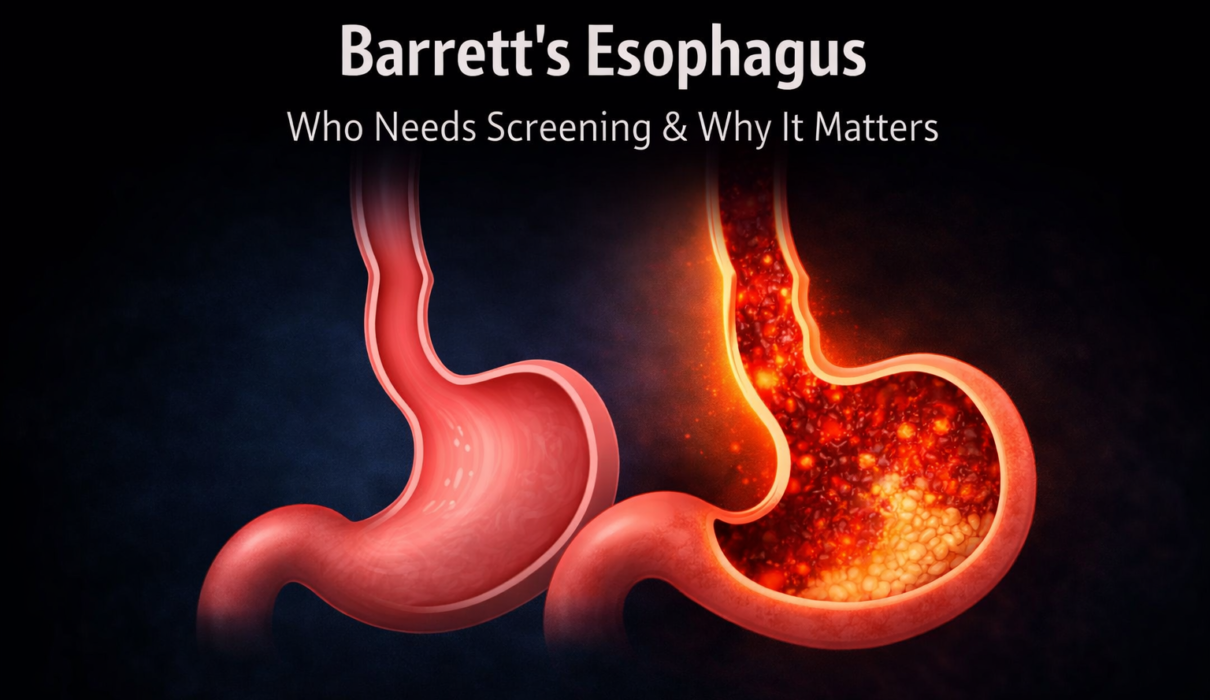
The esophagus is lined with squamous cells, the same type of flat cells that line the inside of your mouth. These cells are well suited to their job of moving food downward but are not designed to tolerate repeated exposure to stomach acid. The stomach, by contrast, is lined with columnar cells that are specifically equipped to handle acid. They produce a protective mucus layer and are built for the acidic environment they live in.
When stomach acid repeatedly contacts the lower esophagus over months and years, the squamous cells in that area begin to be replaced by a type of columnar cell more similar to those found in the intestinal lining. This cellular transformation is called intestinal metaplasia, and the resulting change in the esophageal lining is what is diagnosed as Barrett’s esophagus.
In simple terms, the body is attempting to protect a repeatedly injured area by replacing vulnerable cells with more acid-resistant ones. It is an adaptive response, but it is not a benign one. These new columnar cells, while more resistant to acid, carry a higher risk of developing abnormal changes over time.
Barrett’s esophagus is diagnosed in an estimated 3 to 5 percent of Americans who undergo upper endoscopy, though the true prevalence in the general population is thought to be higher because many people with the condition have never been scoped (Shaheen and Richter, 2009).
How Barrett’s Esophagus Develops
Chronic, inadequately controlled GERD is the primary driver of Barrett’s esophagus. The longer and more severe the acid exposure to the lower esophagus, the greater the likelihood of intestinal metaplasia developing.
Not everyone with GERD develops Barrett’s esophagus, and researchers have not fully established why some people with longstanding reflux develop it while others do not. What is known is that certain factors appear to increase the likelihood.
Long duration of GERD symptoms, particularly more than five years of frequent reflux, is one of the strongest risk factors. Obesity, especially central abdominal obesity, increases the risk both through its mechanical effects on the lower esophageal sphincter and through other metabolic pathways that are still being studied. Hiatal hernia, which disrupts normal LES function and increases acid exposure, is also associated with higher rates of Barrett’s esophagus.
Demographic factors matter as well. Barrett’s esophagus is significantly more common in white males over the age of 50. It is less commonly diagnosed in women and in Black and Hispanic Americans, though it does occur across all demographic groups (American College of Gastroenterology, 2022).
Smoking is an independent risk factor for Barrett’s esophagus beyond its general effect on GERD. Interestingly, alcohol use has not been consistently associated with increased Barrett’s risk despite its role in worsening reflux.
The Cancer Connection: Understanding the Real Risk
Barrett’s esophagus is considered a precancerous condition because it can progress through a sequence of changes toward esophageal adenocarcinoma, a type of cancer that develops in the glandular cells of the lower esophagus.
The progression follows a defined pathway. Barrett’s esophagus without dysplasia, meaning without abnormal cellular changes, is the starting point. From there, low-grade dysplasia can develop, followed by high-grade dysplasia, and in some cases eventually esophageal adenocarcinoma.
Here is where the context that most people never hear becomes important.
The annual rate of progression from non-dysplastic Barrett’s esophagus to esophageal adenocarcinoma is approximately 0.1 to 0.3 percent per year (Hvid-Jensen et al., 2011). That means for every 1,000 people with Barrett’s esophagus who do not yet have dysplasia, roughly one to three will develop cancer in any given year.
Over a lifetime that risk accumulates and is real. But it also means that the vast majority of people with Barrett’s esophagus will never develop esophageal cancer, especially with appropriate monitoring and management. This is a fundamentally different picture than the one most people carry in their heads when they first hear the diagnosis.
The risk increases meaningfully when dysplasia is present, which is precisely why surveillance endoscopy, the regular monitoring of Barrett’s esophagus over time, is so important. Finding dysplasia early allows intervention before cancer develops.
Who Should Be Screened
Screening for Barrett’s esophagus involves upper endoscopy, a procedure in which a thin flexible camera is passed through the mouth into the esophagus and stomach. During the procedure, the gastroenterologist can directly visualize the esophageal lining and take small tissue biopsies from any areas that appear abnormal. The biopsies are then examined under a microscope to confirm the diagnosis and assess whether dysplasia is present.
The American College of Gastroenterology recommends considering screening endoscopy for Barrett’s esophagus in patients with chronic GERD, defined as symptoms occurring at least weekly for five or more years, combined with multiple risk factors (Shaheen et al., 2022).
Those risk factors include being male, being over 50 years old, being white, having obesity particularly with central fat distribution, having a current or past smoking history, and having a first-degree relative with Barrett’s esophagus or esophageal adenocarcinoma.
Women are not excluded from screening consideration, but the lower prevalence of Barrett’s esophagus and esophageal adenocarcinoma in women means the threshold for recommending screening is different and is individualized based on risk factors.
Importantly, not everyone with longstanding GERD needs to be scoped. Screening is a risk-stratified decision made between a patient and their gastroenterologist based on the overall picture of individual risk factors, not simply on the presence of reflux symptoms alone.
It is also worth noting that a significant proportion of people diagnosed with esophageal adenocarcinoma report no prior heartburn symptoms. This means that Barrett’s esophagus and its associated cancer can develop in people who have had silent or atypical GERD without ever experiencing classic heartburn.
What Happens After a Barrett’s Esophagus Diagnosis
If Barrett’s esophagus is found on endoscopy, the management approach depends on whether dysplasia is present and at what grade.
For non-dysplastic Barrett’s esophagus, the standard approach is surveillance endoscopy every three to five years to monitor for any progression. During the surveillance period, optimizing GERD treatment is important, both to reduce ongoing acid exposure to the Barrett’s segment and to manage symptoms. High-dose PPI therapy is commonly used in this setting.
For low-grade dysplasia, the approach has evolved significantly in recent years. Current American guidelines from the American Society for Gastrointestinal Endoscopy and the American College of Gastroenterology favor endoscopic eradication therapy over surveillance alone for most patients with confirmed low-grade dysplasia, given the higher risk of progression compared to non-dysplastic Barrett’s (Shaheen et al., 2022).
For high-grade dysplasia, endoscopic eradication therapy is strongly recommended. The good news is that highly effective, minimally invasive endoscopic techniques now exist to treat Barrett’s esophagus with dysplasia without requiring surgery.
Radiofrequency ablation is the most widely used and well-studied technique. It uses controlled heat energy delivered through an endoscope to destroy the abnormal Barrett’s tissue, allowing normal squamous cells to regrow in its place. Studies have shown eradication rates of over 90 percent for dysplasia and around 80 percent for intestinal metaplasia with this approach (Shaheen et al., 2011).
Endoscopic mucosal resection is another technique used when there are visible nodules or raised areas in the Barrett’s segment that need to be removed and examined pathologically.
Cryotherapy, which uses extreme cold to destroy abnormal tissue, is also available at specialized centers.
These endoscopic approaches have largely replaced surgical esophagectomy for treating dysplastic Barrett’s esophagus in most patients, representing a significant advance in how this condition is managed in the United States.
The Role of Lifestyle in Barrett’s Esophagus
While Barrett’s esophagus itself cannot be reversed through lifestyle changes alone, managing the underlying GERD that drives it remains important even after diagnosis.
Reducing acid exposure through dietary modifications, weight loss, elevating the head of the bed, and avoiding late meals all reduce the ongoing insult to the Barrett’s segment. Optimized GERD management is a standard component of the care plan for Barrett’s esophagus regardless of whether endoscopic treatment is pursued.
Some research has explored whether aspirin or other anti-inflammatory medications might reduce the risk of progression in Barrett’s esophagus, given the role of inflammation in cancer development. The evidence is not yet strong enough to recommend aspirin specifically for Barrett’s surveillance patients outside of clinical trials, but it is an active area of investigation (Jankowski et al., 2018).
Smoking cessation is important for anyone with Barrett’s esophagus, both because smoking is a risk factor for progression and because of its broader health implications.
Frequently Asked Questions
Q: If I have had heartburn for years, does that mean I probably have Barrett’s esophagus? Not necessarily. The majority of people with chronic GERD do not develop Barrett’s esophagus. The condition develops in an estimated 10 to 15 percent of people with chronic reflux, and even within that group the cancer risk is low. Longstanding GERD does warrant a conversation with your gastroenterologist about whether screening is appropriate given your individual risk factors.
Q: Can Barrett’s esophagus go away on its own? Non-dysplastic Barrett’s esophagus does not typically reverse spontaneously. However, with endoscopic treatment such as radiofrequency ablation, the abnormal tissue can be successfully eradicated in the majority of patients. Optimizing GERD management reduces ongoing injury but does not reliably cause established Barrett’s tissue to revert to normal.
Q: Does having Barrett’s esophagus mean I will get esophageal cancer? No. The annual progression rate from non-dysplastic Barrett’s esophagus to cancer is approximately 0.1 to 0.3 percent per year. The vast majority of people with Barrett’s esophagus never develop esophageal cancer, particularly when the condition is monitored appropriately through regular surveillance endoscopy.
Q: Is the endoscopy procedure for Barrett’s screening painful? Upper endoscopy is performed under conscious sedation in the United States, meaning most patients are comfortable throughout and have little or no memory of the procedure afterward. It typically takes 15 to 30 minutes and is done as an outpatient procedure. Most people return to normal activities the same day.
Q: My doctor found Barrett’s esophagus but said no dysplasia is present. What does that mean for me? Non-dysplastic Barrett’s esophagus without any cellular abnormalities carries the lowest risk of progression. The standard recommendation is surveillance endoscopy every three to five years to monitor for any changes, along with optimizing your GERD treatment to reduce ongoing acid exposure. This is a manageable situation that does not require immediate intervention.
Practical Takeaway
Barrett’s esophagus is a real and meaningful complication of chronic GERD that deserves proper attention, not panic. It is a cellular change in the lower esophageal lining caused by repeated acid exposure, and it does carry an increased risk of esophageal adenocarcinoma compared to the general population. But the absolute annual risk of cancer progression in non-dysplastic Barrett’s esophagus is low, and regular surveillance endoscopy allows any progression toward dysplasia to be caught and treated early.
Screening is recommended for patients with long-duration GERD and multiple risk factors, not for everyone with occasional heartburn. If Barrett’s esophagus is found, the management approach is guided by whether dysplasia is present, with highly effective endoscopic treatment options available for those who need intervention.
Disclaimer:This article is for educational purposes only and does not constitute medical advice. Always consult a qualified healthcare professional for personal health concerns.
Final Thoughts
A Barrett’s esophagus diagnosis is not a cancer diagnosis. It is a signal that the esophagus has been under significant acid stress, that monitoring is now important, and that the underlying GERD driving it deserves more serious management than over-the-counter antacids alone can provide.
Understanding what the condition actually is, what the risks actually are, and what modern American gastroenterology can do about it turns an anxiety-inducing diagnosis into a manageable one. That understanding starts here.
For the complete picture of GERD including causes, symptoms, and treatment options, visit our GERD and Acid Reflux Explained guide.
References
Shaheen NJ, Richter JE. Barrett’s oesophagus. Lancet. 2009;373(9666):850–861. https://pubmed.ncbi.nlm.nih.gov/19269522
Hvid-Jensen F, Pedersen L, Drewes AM, Sorensen HT, Funch-Jensen P. Incidence of adenocarcinoma among patients with Barrett’s esophagus. N Engl J Med. 2011;365(15):1375–1383. https://pubmed.ncbi.nlm.nih.gov/21995385
Shaheen NJ, Falk GW, Iyer PG, et al. Diagnosis and management of Barrett’s esophagus: an updated ACG guideline. Am J Gastroenterol. 2022;117(4):559–587. https://pubmed.ncbi.nlm.nih.gov/35354777
Shaheen NJ, Sharma P, Overholt BF, et al. Radiofrequency ablation in Barrett’s esophagus with dysplasia. N Engl J Med. 2009;360(22):2277–2288. https://pubmed.ncbi.nlm.nih.gov/19474425
Jankowski JAZ, de Caestecker J, Love SB, et al. Esomeprazole and aspirin in Barrett’s oesophagus. Lancet. 2018;392(10145):400–408. https://pubmed.ncbi.nlm.nih.gov/30057104
American College of Gastroenterology. Barrett’s Esophagus. 2022. https://gi.org/topics/barretts-esophagus/