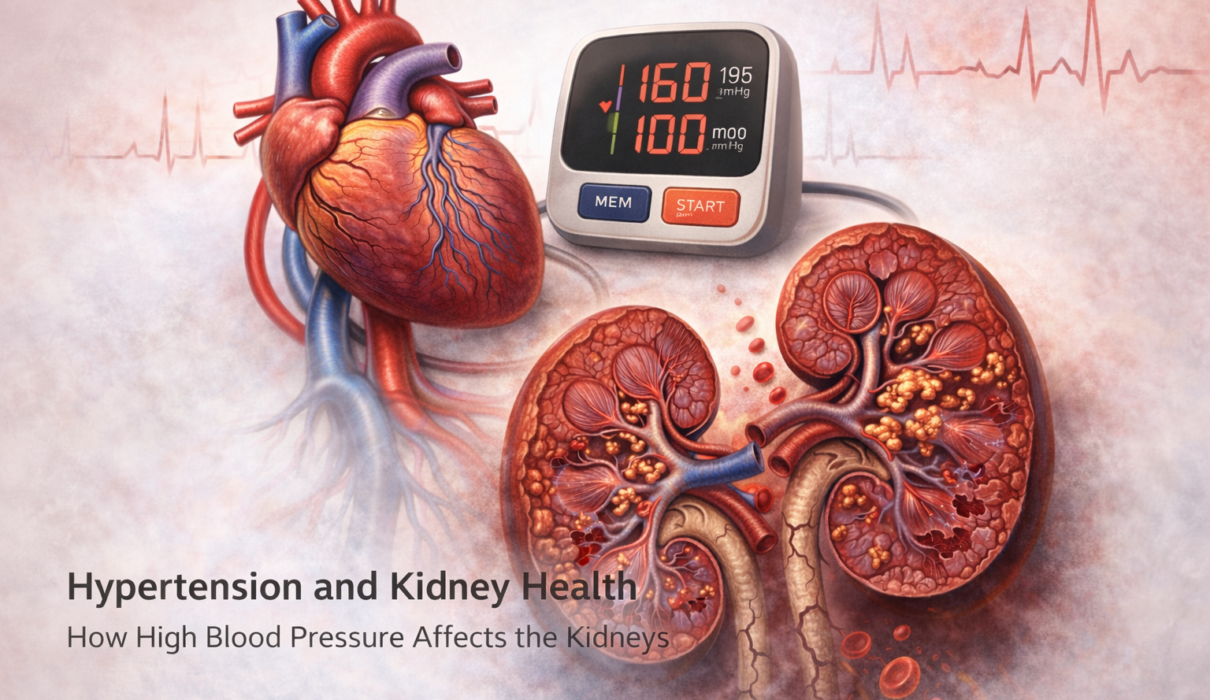
Hypertension and kidney disease have one of the most clinically important relationships in medicine – and it runs in both directions. High blood pressure damages the kidneys over time. Damaged kidneys raise blood pressure further. Each condition accelerates the other, and without intervention, the cycle leads progressively toward kidney failure and cardiovascular disease.
In the United States, hypertension is the second leading cause of kidney failure after diabetes, accounting for roughly 25% of all end-stage renal disease cases. Yet the kidney damage caused by high blood pressure almost never announces itself with symptoms – it’s detected on lab tests, often years into its progression.
This article explains how hypertension damages the kidneys, how kidney disease raises blood pressure, what the warning signs on lab tests look like, and what treatment approaches actually protect kidney function.
How the Kidneys Regulate Blood Pressure
Before getting into damage, it’s worth understanding why kidneys and blood pressure are so fundamentally connected.
The kidneys control blood pressure through three major mechanisms:
Fluid volume regulation: By controlling how much sodium and water is retained or excreted, the kidneys directly regulate blood volume. More volume means more pressure. This is why salt intake affects blood pressure – the kidneys have to handle the extra sodium load, and in many people (particularly older adults and Black Americans who tend to be more salt-sensitive), this raises blood pressure.
The renin-angiotensin-aldosterone system (RAAS): When the kidneys sense reduced blood flow – either because blood pressure is low or because they’re being underperfused – they release renin, which triggers a hormonal cascade that produces angiotensin II. Angiotensin II is one of the most potent vasoconstrictors in the body – it tightens blood vessels, raises blood pressure, and signals the adrenal glands to release aldosterone, which causes the kidneys to retain more sodium and water. This system is designed for short-term blood pressure rescue, but in hypertension and kidney disease, it gets chronically overactivated.
Erythropoietin and other hormones: The kidneys produce hormones involved in red blood cell production and vascular tone. As kidney function declines, these regulatory functions are impaired, contributing to anemia and further blood pressure instability.
How Hypertension Damages the Kidneys
Each kidney contains approximately one million filtering units called nephrons, each consisting of a tiny capillary tuft (glomerulus) surrounded by a collection structure (Bowman’s capsule) that channels filtered fluid into the tubule system. The glomerulus is where the actual filtration happens – small molecules including waste products pass through while large proteins and blood cells are retained.
When blood pressure is chronically elevated, the glomeruli bear the brunt of it. The increased pressure forces blood through the filtering membrane with more force than the delicate capillaries are designed to handle. Over time this causes:
Glomerulosclerosis: Scarring and stiffening of the glomerular capillaries. Scarred glomeruli can’t filter effectively and are progressively replaced by fibrous tissue. As more nephrons scar over, filtration capacity falls – this is reflected in a declining eGFR.
Arteriosclerosis of renal arterioles: The small arteries feeding each glomerulus (afferent and efferent arterioles) thicken and stiffen in response to sustained pressure. This reduces blood flow to the nephrons and impairs autoregulation – the kidney’s normal ability to maintain stable glomerular pressure despite fluctuations in systemic blood pressure.
Proteinuria: As the glomerular filtration barrier is damaged, it becomes leaky. Albumin and other proteins that should be retained in the blood spill into the urine. Proteinuria isn’t just a marker of damage – it’s itself toxic to tubular cells and accelerates further kidney decline.
Tubular injury: Reduced blood flow and ischemia from arteriosclerosis injures the tubular cells responsible for reabsorption and secretion. This impairs the kidney’s ability to concentrate urine, handle electrolytes, and regulate acid-base balance.
This pattern of injury – glomerulosclerosis, arteriosclerosis, and tubular atrophy – is called hypertensive nephrosclerosis, and it’s one of the most common causes of chronic kidney disease in adults over 60.
The insidious feature of hypertensive kidney damage is that it’s entirely asymptomatic until a substantial proportion of nephron mass has been lost. The kidneys have enormous reserve capacity – you can lose 50-60% of nephron function before serum creatinine begins to rise meaningfully above normal. By the time most people notice anything, significant structural damage has already occurred.
The Feedback Loop: How Kidney Disease Raises Blood Pressure
Once kidney damage begins, the situation compounds. Damaged kidneys:
- Retain more sodium and water, raising blood volume and therefore blood pressure
- Overactivate the RAAS – scarred, ischemic kidney tissue chronically releases renin even when it shouldn’t, driving persistent vasoconstriction
- Produce less of the vasodilatory hormones that normally help keep blood pressure in check
- Eventually lose the ability to excrete the extra fluid load that accumulates
The result is a vicious cycle: hypertension damages the kidneys → damaged kidneys impair blood pressure regulation → blood pressure rises further → more kidney damage occurs. This is why hypertension and CKD so frequently coexist and why patients with both conditions are significantly harder to treat than those with either alone.
What the Lab Tests Show – and When to Look for Them
Because hypertensive kidney damage is silent, detection depends entirely on testing. The key markers:
Serum creatinine and eGFR: Creatinine rises as filtration declines. eGFR – the estimated glomerular filtration rate – converts creatinine into an estimate of filtration capacity adjusted for age, sex, and body size. A persistently low eGFR (below 60 mL/min/1.73m² on two measurements taken more than three months apart) meets the KDIGO criteria for chronic kidney disease.
Urine albumin-to-creatinine ratio (UACR): This is often the first detectable sign of hypertensive kidney damage – it can be abnormal years before eGFR begins to fall. A UACR above 30 mg/g (microalbuminuria) on two or more tests indicates early kidney damage. Above 300 mg/g (macroalbuminuria) signals significant filtration barrier injury.
Blood pressure monitoring at home: Nighttime blood pressure – which should dip during sleep – is particularly relevant for kidney health. “Non-dipping” blood pressure patterns (where nocturnal BP doesn’t fall as expected) are associated with greater kidney damage progression than daytime readings alone suggest.
Reference points for monitoring:
| Marker | Target / Action Threshold |
|---|---|
| eGFR above 60 | Normal to mild decrease; monitor annually |
| eGFR 30-60 | CKD Stage 3; nephrology referral consideration |
| eGFR below 30 | CKD Stage 4-5; nephrology referral essential |
| UACR below 30 mg/g | Normal |
| UACR 30-300 mg/g | Early damage; intensify BP treatment |
| UACR above 300 mg/g | Significant damage; ACE inhibitor / ARB therapy essential |
The NIDDK and American Diabetes Association both recommend annual testing of eGFR and UACR in people with hypertension, particularly when diabetes is also present.
How Treatment Protects the Kidneys
Blood pressure targets in CKD: The 2021 KDIGO guidelines recommend a blood pressure target of below 120 mmHg systolic (using standardized measurement) for most adults with CKD, aligning with SPRINT trial targets. For people with significant proteinuria, aggressive blood pressure lowering is particularly important because elevated pressure directly drives further proteinuria and fibrosis.
ACE inhibitors and ARBs – the kidney-protective drugs: Angiotensin-converting enzyme (ACE) inhibitors (lisinopril, enalapril, ramipril) and angiotensin receptor blockers (ARBs) (losartan, valsartan, irbesartan) are first-line antihypertensive therapy in people with CKD and proteinuria for two reasons:
First, they lower blood pressure. Second, and critically, they reduce intraglomerular pressure independently of their systemic blood pressure effect – by dilating the efferent arteriole, they reduce the pressure inside the glomerular capillary itself, slowing damage even beyond what blood pressure reduction alone achieves. Multiple landmark trials – RENAAL, IDNT, AASK – have demonstrated that ACE inhibitors and ARBs slow the progression of kidney disease in hypertensive nephropathy and diabetic nephropathy, reducing the risk of doubling serum creatinine or reaching kidney failure.
These two drug classes should not be combined (dual blockade is associated with worse outcomes and hyperkalemia), but either alone is superior to other antihypertensive classes specifically for kidney protection in the presence of proteinuria.
SGLT2 inhibitors: A newer class of drugs originally developed for type 2 diabetes (canagliflozin, dapagliflozin, empagliflozin) has demonstrated compelling kidney-protective effects independent of blood sugar lowering. The CREDENCE and DAPA-CKD trials showed significant reduction in kidney failure, cardiovascular events, and death in patients with CKD and proteinuria treated with SGLT2 inhibitors. Current KDIGO and ADA guidelines now recommend them for most patients with CKD and proteinuria regardless of diabetes status.
Dietary sodium restriction: Reducing sodium intake to below 2,300 mg per day (with a target of 1,500-2,000 mg for those with CKD) lowers blood pressure and also directly reduces glomerular hyperfiltration and proteinuria. The DASH diet, which emphasizes this alongside high potassium and low saturated fat, has demonstrated blood pressure and kidney protection benefits in clinical trials.
Note on potassium: People with advanced CKD (particularly eGFR below 30) have impaired potassium excretion and are at risk for hyperkalemia, which can cause dangerous cardiac arrhythmias. ACE inhibitors and ARBs also raise potassium. Potassium levels need regular monitoring in this population, and dietary potassium restriction may be necessary.
Who Is at Greatest Risk
Hypertensive kidney damage is more likely and progresses faster in:
- People with both hypertension and diabetes – the combination is far more damaging than either alone
- Black Americans – who have a 3-4 times higher rate of hypertension-related kidney failure than white Americans, partly due to higher rates of salt sensitivity and a higher prevalence of a genetic variant (APOL1) that increases susceptibility to kidney disease
- People with longstanding poorly controlled hypertension – duration and degree of elevation both matter
- People with metabolic syndrome or obesity
- Smokers – smoking accelerates kidney damage through endothelial injury and reduced renal blood flow
- Those who regularly use NSAIDs (ibuprofen, naproxen) – these drugs reduce renal blood flow and can precipitate acute kidney injury, particularly in people with existing kidney disease or volume depletion
Frequently Asked Questions
My blood pressure is controlled on medication. Are my kidneys safe? Better controlled, not necessarily safe. Antihypertensive treatment significantly slows kidney damage progression but may not fully halt it, particularly in the presence of proteinuria or long-standing prior damage. Annual monitoring of eGFR and UACR remains important even with well-controlled blood pressure.
Can kidney damage from hypertension be reversed? Partial improvement is possible, particularly in early stages. When proteinuria is reduced through ACE inhibitor/ARB therapy and blood pressure is well controlled, some degree of glomerular healing can occur. Established glomerulosclerosis (scarring) doesn’t reverse, but slowing further damage is achievable and meaningful – even modest slowing of decline can add years before kidney failure.
Should I avoid ibuprofen and naproxen if I have hypertension? If you have both hypertension and any degree of CKD, regular NSAID use should be avoided and even occasional use discussed with your doctor. NSAIDs constrict the afferent arteriole and reduce renal blood flow, impairing filtration and potentially causing acute kidney injury on top of chronic disease. Acetaminophen is generally safer for pain relief in people with kidney disease.
What symptoms indicate my kidneys might be failing? The frustrating answer is: often none until late stages. Fatigue, decreased urine output, frothy urine (from heavy proteinuria), swelling in the ankles and legs, and high blood pressure that becomes harder to control can all occur – but these typically appear only after significant function is already lost. This is precisely why lab monitoring, not symptom tracking, is the right tool for people with hypertension.
At what point does a nephrologist need to be involved? Most guidelines suggest nephrology referral when eGFR falls below 30 mL/min/1.73m², when there’s significant proteinuria (UACR above 300 mg/g) that isn’t responding to treatment, when there’s uncertainty about the cause of kidney disease, or when blood pressure is difficult to control. Earlier referral is appropriate for rapidly declining eGFR regardless of absolute level.
Disclaimer
This article is for educational purposes only and does not constitute medical advice. Kidney health monitoring and hypertension management in the context of CKD should be directed by a qualified healthcare provider. Do not adjust medications or dietary practices based on this content without consulting your doctor.
References
- KDIGO 2021 Clinical Practice Guideline for the Management of Blood Pressure in Chronic Kidney Disease. Kidney International. 2021;99(3S):S1-S87. https://doi.org/10.1016/j.kint.2020.11.003
- Whelton PK, et al. 2017 ACC/AHA high blood pressure guideline. Journal of the American College of Cardiology. 2018;71(19):e127-e248. https://doi.org/10.1016/j.jacc.2017.11.006
- Brenner BM, et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy (RENAAL trial). New England Journal of Medicine. 2001;345(12):861-869. https://doi.org/10.1056/NEJMoa011161
- Perkovic V, et al. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy (CREDENCE trial). New England Journal of Medicine. 2019;380(24):2295-2306. https://doi.org/10.1056/NEJMoa1811744
- National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). High blood pressure and kidney disease. https://www.niddk.nih.gov/health-information/kidney-disease/high-blood-pressure
- Centers for Disease Control and Prevention (CDC). Chronic kidney disease and hypertension. https://www.cdc.gov/kidneydisease/index.html
- National Kidney Foundation. High blood pressure and kidney disease. https://www.kidney.org/kidney-topics/high-blood-pressure
- Heerspink HJL, et al. Dapagliflozin in patients with chronic kidney disease (DAPA-CKD). New England Journal of Medicine. 2020;383(15):1436-1446. https://doi.org/10.1056/NEJMoa2024816
- Saran R, et al. US Renal Data System 2022 Annual Data Report. American Journal of Kidney Diseases. 2023;81(3S1):A8-A12. https://doi.org/10.1053/j.ajkd.2022.12.001
- Levin A, et al. Global kidney health 2017 and beyond: a roadmap for closing gaps in care, research, and policy. The Lancet. 2017;390(10105):1888-1917. https://doi.org/10.1016/S0140-6736(17)30788-2