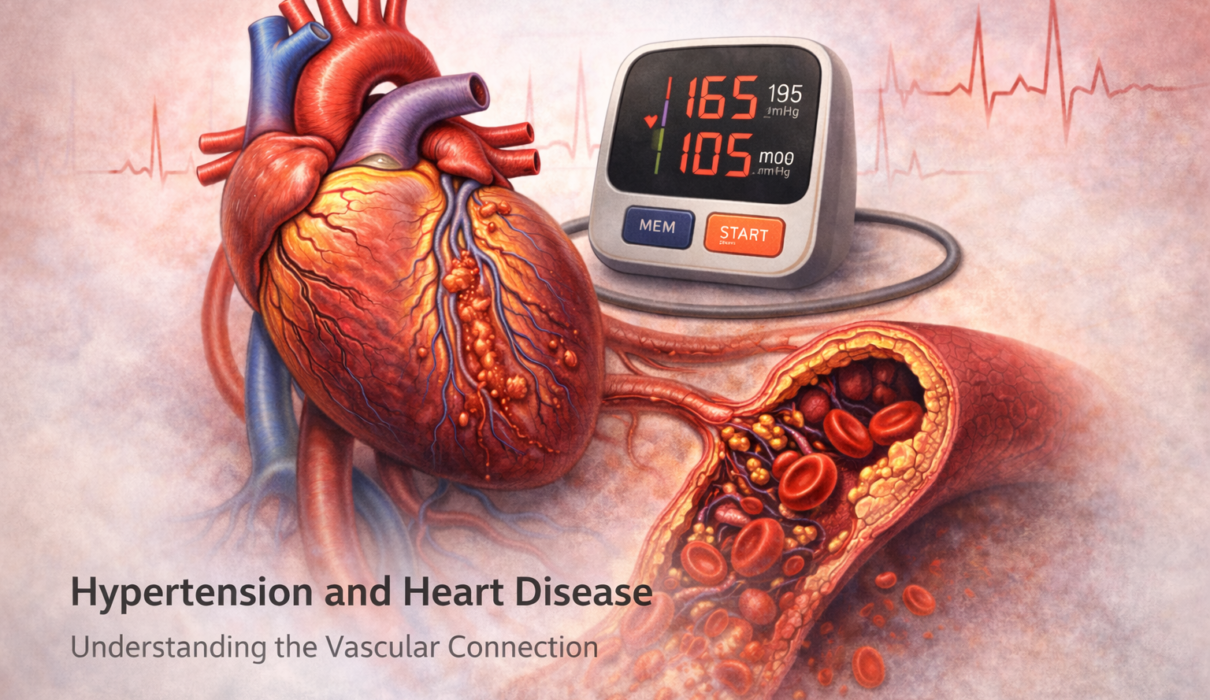
High blood pressure and heart disease don’t just happen to occur together. One causes the other – and over time, each makes the other worse. Nearly half of Americans with hypertension will develop some form of heart disease related to it, yet the damage happens so gradually and so quietly that most people have no idea it’s occurring until something goes seriously wrong.
This article explains exactly how sustained high blood pressure injures the heart and blood vessels, what structural changes develop over years of uncontrolled pressure, which specific heart conditions hypertension drives, and what the risk numbers actually look like.
The Fundamental Mechanism: Pressure as a Physical Force
To understand how hypertension harms the heart, it helps to think of blood pressure as a physical force that arteries and heart muscle experience with every single beat – roughly 100,000 times a day.
When that force is consistently elevated, two things happen in parallel. First, the arteries themselves are stressed and damaged – the inner lining (endothelium) gets injured, triggering an inflammatory response that accelerates plaque formation. Second, the heart muscle has to work against higher resistance with every pump – the cardiac equivalent of constantly lifting a heavier weight than it’s designed for.
Both processes unfold silently, typically over years or decades, long before any symptoms develop.
Left Ventricular Hypertrophy: The Heart Remodeling Under Pressure
The left ventricle is the main pumping chamber of the heart – the one responsible for pushing blood out into the aorta and then throughout the body. When blood pressure is chronically elevated, the left ventricle has to generate more force with every contraction to overcome the increased arterial resistance.
The heart responds to this sustained overload the same way a skeletal muscle responds to consistent heavy exercise: it gets bigger. The wall of the left ventricle thickens – a condition called left ventricular hypertrophy (LVH).
Initially this seems like an adaptive response. A thicker muscle can generate more force. But LVH has serious consequences:
- The thickened muscle becomes stiffer and less compliant, making it harder for the ventricle to relax and fill properly between beats – a problem called diastolic dysfunction
- As the muscle mass increases, the blood supply to that muscle needs to increase too, but the coronary arteries can’t always keep up, leaving the enlarged heart partially ischemic
- The thickened, stiff muscle creates electrical instability that predisposes to dangerous arrhythmias
LVH is detectable on an ECG or echocardiogram and is one of the most important early signs that hypertension is damaging the heart. According to the Framingham Heart Study – the landmark decades-long cardiovascular study from which much of our understanding of heart disease risk derives – LVH increases the risk of heart failure, myocardial infarction, sudden cardiac death, and stroke independently of other risk factors.
LVH is present in roughly 15-20% of people with hypertension – and most of them have no symptoms at the time it’s discovered. It’s a structural change that develops in plain sight but without any warning signal.
Heart Failure: When the Pump Finally Gives Out
Heart failure is one of the most feared cardiovascular complications of hypertension, and it’s more common than most people realize. Hypertension is the single most common risk factor for heart failure in the United States, accounting for approximately 75% of cases, according to the American Heart Association.
Heart failure doesn’t mean the heart stops – it means the heart can no longer pump blood efficiently enough to meet the body’s needs. Hypertension drives heart failure through two distinct mechanisms:
Heart failure with preserved ejection fraction (HFpEF): The left ventricle becomes stiff and noncompliant from years of pressure overload and LVH. It can still squeeze adequately, but it can’t relax properly between beats to fill with blood – so despite a normal squeeze fraction (ejection fraction), the output is inadequate. HFpEF is the dominant form of heart failure in older adults with longstanding hypertension, particularly women. It’s also notoriously harder to treat than the other type.
Heart failure with reduced ejection fraction (HFrEF): After years of pressure overload, the ventricle eventually dilates and the muscle thins and weakens. Squeeze function declines. This is the “classic” image of heart failure most people have, and it’s also driven by the coronary artery disease that hypertension accelerates.
Symptoms of heart failure – shortness of breath with exertion or lying flat, ankle swelling, fatigue, reduced exercise tolerance – typically emerge only after significant structural damage has already occurred.
Coronary Artery Disease: Atherosclerosis Accelerated
The coronary arteries are the blood vessels that supply oxygen to the heart muscle itself. When these narrow from atherosclerotic plaque, the heart muscle doesn’t get enough blood during exertion – causing chest pain (angina) – or gets no blood at all when a plaque ruptures and a clot forms – causing a heart attack.
Hypertension accelerates atherosclerosis through several overlapping mechanisms:
- Elevated pressure physically injures the endothelium (the inner lining of arteries), making it more permeable to LDL cholesterol particles that then deposit in the arterial wall
- Endothelial injury triggers chronic inflammation – macrophages accumulate, foam cells form, and the inflammatory atherosclerotic plaque grows
- Pressure-related wall stress promotes oxidative stress and reduces nitric oxide availability, impairing the artery’s ability to dilate
- Hypertension combined with other risk factors – diabetes, high LDL, smoking – creates a multiplicative rather than additive effect on coronary risk
The SPRINT trial (Systolic Blood Pressure Intervention Trial), a landmark NIH-funded study published in 2015, demonstrated that treating systolic blood pressure to below 120 mmHg rather than the then-standard below 140 mmHg reduced major cardiovascular events by 25% and cardiovascular death by 43%. This underscored how directly blood pressure control translates to reduced heart attack and heart failure risk.
People with hypertension have approximately twice the risk of coronary artery disease compared to those with normal blood pressure. When hypertension coexists with diabetes, the risk is four to five times higher.
Atrial Fibrillation: The Rhythm Disruption
Atrial fibrillation (AFib) – an irregular, often rapid heart rhythm originating in the upper chambers of the heart – is the most common sustained cardiac arrhythmia, affecting more than 6 million Americans. Hypertension is its single most common modifiable risk factor.
The pathway from hypertension to AFib runs through the structural changes already described. As the left ventricle stiffens from LVH and diastolic dysfunction, pressure backs up into the left atrium. The atrium dilates and its wall undergoes fibrotic change – creating the electrical disorganization that drives AFib.
AFib matters enormously for stroke risk. The irregular rhythm allows blood to pool in the left atrial appendage and form clots. When a clot dislodges, it can travel to the brain and cause a stroke. People with AFib have a five-fold increased stroke risk compared to those without it – and the strokes caused by AFib tend to be larger and more disabling than strokes from other causes.
This is why blood pressure control isn’t just about preventing heart attacks – it’s about preventing AFib, which then prevents the strokes that come from it.
Aortic Aneurysm and Hypertensive Emergency
Two additional cardiovascular consequences of hypertension deserve mention:
Aortic aneurysm and dissection: The aorta – the main artery carrying blood from the heart to the rest of the body – bears the highest pressure in the circulatory system. Sustained hypertension weakens the aortic wall, leading to gradual dilation (aneurysm) or, in acute severe hypertension, tearing of the layers of the aortic wall (dissection). Aortic dissection is a medical emergency with high mortality.
Hypertensive emergency: When blood pressure rises acutely to very high levels (typically systolic above 180 mmHg with evidence of organ damage), it can trigger a cascade of acute heart, brain, kidney, and vascular complications. This is distinct from hypertensive urgency (similarly high readings without organ damage) and requires immediate treatment in a hospital setting.
The Risk Reduction Math: Why Blood Pressure Control Works
The relationship between blood pressure and cardiovascular risk is continuous – there’s no magic threshold below which there’s zero extra risk. But the reduction in risk achievable through treatment is substantial and well-documented:
- Each 10 mmHg reduction in systolic blood pressure is associated with approximately 20-25% reduction in major cardiovascular events, 35% reduction in stroke risk, and 40% reduction in heart failure risk, according to meta-analyses of blood pressure-lowering trials published in The Lancet
- Getting from Stage 2 hypertension (≥140/90) to controlled blood pressure (<130/80) reduces 10-year cardiovascular risk by a clinically meaningful margin in virtually every risk category
- The benefits of treatment extend to older adults – the SPRINT trial included adults over 75 and showed significant benefit from intensive blood pressure lowering even in this age group
This is why hypertension management isn’t about a number on a monitor. It’s about the years of accumulated arterial damage that those numbers represent.
Frequently Asked Questions
Can the heart damage from hypertension be reversed? Partially. LVH can regress with effective blood pressure treatment – studies show that ACE inhibitors and ARBs are particularly effective at reducing left ventricular mass alongside blood pressure reduction. Diastolic dysfunction can improve. However, established fibrosis, significant coronary artery disease, and moderate-to-severe heart failure represent damage that doesn’t fully reverse. This is why early treatment before structural changes become entrenched matters so much.
Does having a normal ECG mean my heart is unaffected by hypertension? Not necessarily. A standard 12-lead ECG is relatively insensitive for detecting early LVH – it misses about half of cases that would be apparent on echocardiogram. An echocardiogram (cardiac ultrasound) is the gold standard for assessing whether hypertension has produced structural heart changes. If you’ve had longstanding hypertension, ask your doctor whether an echocardiogram is appropriate.
My blood pressure is controlled on medication. Am I still at risk? Controlled hypertension on medication carries lower risk than uncontrolled hypertension, but some residual risk remains above that of someone who never had elevated pressure. This is partly because treatment begins after some degree of vascular aging has already occurred, and partly because blood pressure control is rarely perfect across all time points. That said, effective treatment produces substantial risk reduction – the goal is consistent control, not perfection.
Can young people with hypertension develop heart disease? Yes. The vascular and cardiac changes from hypertension are cumulative over time, which means someone who develops hypertension at 30 has more years of arterial exposure than someone diagnosed at 55. Studies show that blood pressure in early adulthood predicts cardiovascular risk decades later even when blood pressure normalizes in middle age. Early diagnosis and treatment carry proportionally greater long-term benefit.
What’s the difference between hypertensive heart disease and coronary artery disease? Hypertensive heart disease refers specifically to the structural changes caused by pressure overload – LVH, diastolic dysfunction, and heart failure. Coronary artery disease refers to atherosclerotic narrowing of the coronary arteries. Hypertension contributes to both, but through somewhat different mechanisms – the former through mechanical pressure overload on the heart muscle, the latter through endothelial injury and accelerated atherosclerosis. In practice, both often coexist in the same patient with longstanding hypertension.
Disclaimer
This article is for educational purposes only and does not constitute medical advice. Hypertension management and cardiovascular risk assessment should be individualized by a qualified healthcare provider. Do not start, stop, or change any medications based on this content without medical supervision.
References
- Whelton PK, et al. 2017 ACC/AHA high blood pressure guideline. Journal of the American College of Cardiology. 2018;71(19):e127-e248. https://doi.org/10.1016/j.jacc.2017.11.006
- SPRINT Research Group. A randomized trial of intensive versus standard blood-pressure control. New England Journal of Medicine. 2015;373(22):2103-2116. https://doi.org/10.1056/NEJMoa1511939
- Lewington S, Clarke R, Qizilbash N, et al. Age-specific relevance of usual blood pressure to vascular mortality. The Lancet. 2002;360(9349):1903-1913. https://doi.org/10.1016/S0140-6736(02)11911-8
- Lip GYH, et al. Hypertension and cardiac arrhythmias. European Heart Journal – Cardiovascular Pharmacotherapy. 2017;3(2):108-119. https://doi.org/10.1093/ehjcvp/pvw042
- American Heart Association. Heart failure and high blood pressure. https://www.heart.org/en/health-topics/heart-failure
- Kannel WB. Left ventricular hypertrophy as a risk factor in arterial hypertension. European Heart Journal. 1992;13(Suppl D):82-88. https://doi.org/10.1093/eurheartj/13.suppl_D.82
- Thomopoulos C, Parati G, Zanchetti A. Effects of blood pressure-lowering on outcome incidence in hypertension. Journal of Hypertension. 2014;32(12):2285-2295. https://doi.org/10.1097/HJH.0000000000000378
- National Heart, Lung, and Blood Institute (NHLBI). High blood pressure and heart disease. https://www.nhlbi.nih.gov/health/high-blood-pressure
- Centers for Disease Control and Prevention (CDC). High blood pressure and heart disease. https://www.cdc.gov/heartdisease/high_blood_pressure.htm
- Messerli FH, Rimoldi SF, Bangalore S. The transition from hypertension to heart failure. JACC: Heart Failure. 2017;5(8):543-551. https://doi.org/10.1016/j.jchf.2017.04.012






1 Comment