When women with PCOS ask why their symptoms are so wide-ranging – why a hormonal condition affects their weight, their energy, their skin, their mood, their cardiovascular risk, and their metabolism all at once – the answer involves something that rarely comes up in a typical appointment: chronic low-grade inflammation.
Inflammation is not the most talked-about feature of PCOS. It doesn’t have its own symptom the way irregular periods or acne does. You can’t feel it happening. Most routine blood tests don’t specifically measure it unless a provider thinks to look. But the research increasingly points to chronic low-grade inflammation as one of the central mechanisms running underneath PCOS – interacting with and amplifying the insulin resistance and androgen excess that drive so many of its features.
Understanding inflammation in PCOS doesn’t just explain why the condition is so multisystemic. It also points toward specific, evidence-based interventions that address the inflammatory component directly – and produces improvements across multiple PCOS features simultaneously.
This article is part of our PCOS series. For the full overview of the condition, visit our PCOS Explained guide.
What Chronic Low-Grade Inflammation Actually Is
Before getting into PCOS specifically, it helps to be clear about what kind of inflammation we’re talking about – because “inflammation” means something very different in different contexts.
Acute inflammation is the familiar kind – the redness, swelling, warmth, and pain around an infected wound or a sprained ankle. It is protective, targeted, and self-limiting. The immune system recognizes a threat, mounts a response, resolves the problem, and stands down.
Chronic low-grade inflammation is completely different. It is subtle, persistent, and systemic. There is no visible swelling or obvious pain. The immune system is in a state of low-level continuous activation – producing inflammatory signaling molecules called cytokines at a level that is elevated above normal but not dramatically so. It is detectable through blood markers – most commonly C-reactive protein (CRP), interleukin-6 (IL-6), and tumor necrosis factor-alpha (TNF-alpha) – but produces no obvious symptoms on its own.
What it does produce, over time, is metabolic and vascular damage. Chronic low-grade inflammation is a central driver of insulin resistance, atherosclerosis, type 2 diabetes, and cardiovascular disease. It is one of the reasons why conditions that involve chronic inflammation carry long-term health risks that extend well beyond their obvious presenting features.
“Chronic low-grade inflammation in PCOS is not the kind you can feel. It is a persistent, low-level immune activation that silently drives insulin resistance, worsens androgen excess, and increases cardiovascular risk – operating in the background of every other PCOS feature.”
The Evidence That PCOS Involves Chronic Inflammation
The association between PCOS and elevated inflammatory markers is well-established in the research literature.
Studies consistently find that women with PCOS have elevated levels of:
- C-reactive protein (CRP) – the most widely used clinical marker of systemic inflammation; elevated in PCOS independent of body weight
- Interleukin-6 (IL-6) – a pro-inflammatory cytokine that drives CRP production and promotes insulin resistance
- Tumor necrosis factor-alpha (TNF-alpha) – a cytokine that impairs insulin receptor signaling and promotes fat cell dysfunction
- Monocyte chemoattractant protein-1 (MCP-1) – involved in recruiting immune cells to adipose tissue
- White blood cell count – modestly elevated in PCOS compared to controls
Crucially, elevated inflammatory markers in PCOS are found in both overweight and lean women – confirming that the inflammation is driven by the hormonal and metabolic features of PCOS itself rather than simply by excess body weight (González, 2012). This is the same pattern seen with insulin resistance in PCOS – present across the body weight spectrum, not limited to women who are overweight.
A 2012 review by González – one of the most cited papers in this area – concluded that chronic inflammation is a fundamental feature of PCOS that may play a central role in its pathogenesis and long-term health consequences, not merely a secondary association.
How Inflammation Interacts With PCOS: The Mechanisms
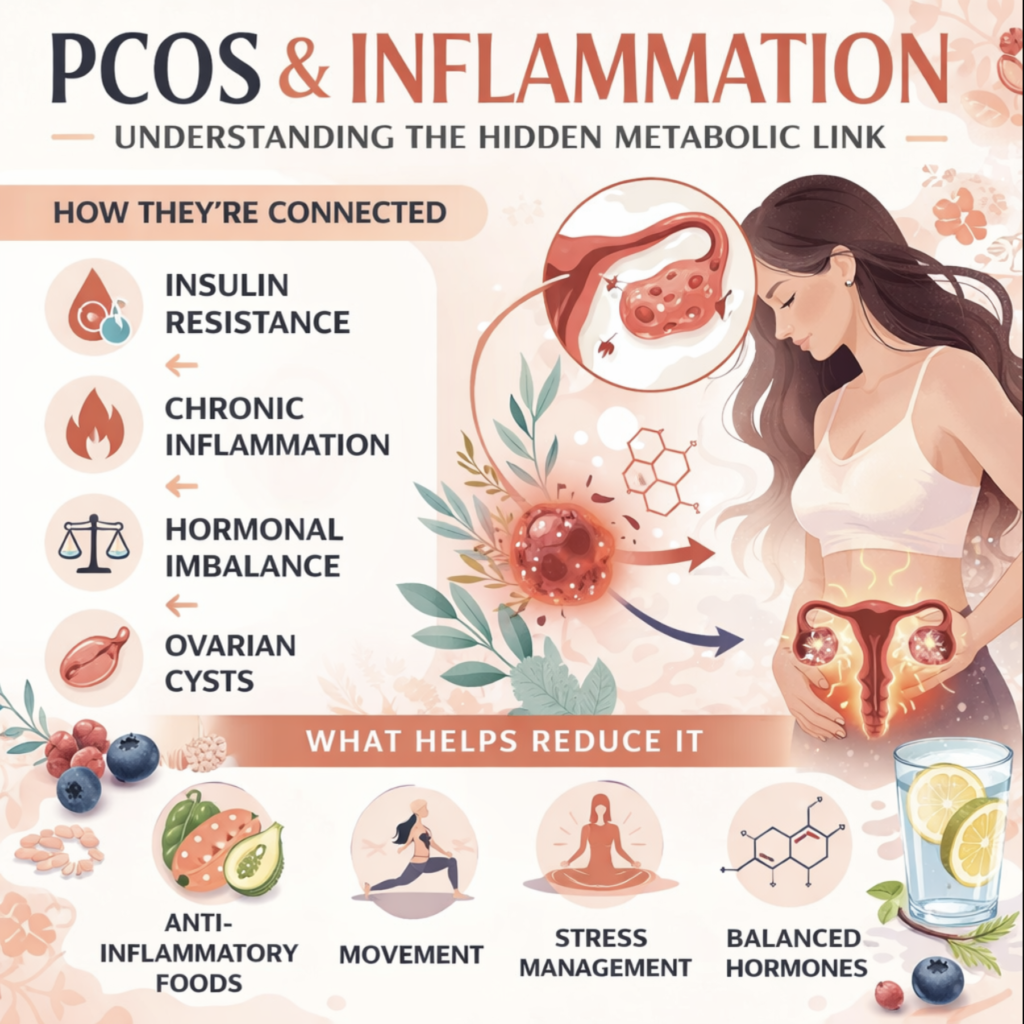
The relationship between inflammation and PCOS is not one-directional. It forms a network of reinforcing connections where inflammation worsens PCOS features and PCOS features worsen inflammation.
Inflammation and Insulin Resistance
This is the most important and best-established connection. Inflammatory cytokines – particularly TNF-alpha and IL-6 – directly interfere with insulin receptor signaling. They activate intracellular pathways (specifically IRS-1 serine phosphorylation) that impair the insulin receptor’s ability to transmit its signal into the cell. The result is reduced cellular glucose uptake – in other words, worsened insulin resistance.
In PCOS, where insulin resistance is already present in 65 to 70 percent of women, chronic inflammation amplifies the problem and makes it harder to correct.
The reverse is also true: insulin resistance promotes inflammation. Elevated insulin levels stimulate the production of pro-inflammatory cytokines, creating a cycle where each side worsens the other.
Inflammation and Androgen Excess
Inflammatory markers correlate with androgen levels in PCOS. This is not a coincidence – there are direct mechanistic connections:
- Inflammatory cytokines stimulate ovarian androgen production, contributing to hyperandrogenism
- Elevated androgens themselves promote inflammatory signaling in adipose tissue and elsewhere
- The combination of elevated insulin and elevated inflammatory cytokines creates a particularly potent stimulus for ovarian androgen overproduction
This means that addressing inflammation is not just a cardiovascular health measure in PCOS – it has potential to improve the hormonal features of the condition directly.
Inflammation and Adipose Tissue
Adipose tissue – particularly visceral fat around the abdominal organs – is not metabolically inert. It is an active endocrine organ that produces cytokines and inflammatory mediators. In women with PCOS, who tend to have disproportionate central adiposity driven by androgen excess, adipose tissue becomes a significant source of the chronic inflammation observed in the condition.
This creates a further reinforcing cycle: PCOS promotes central fat storage through androgens, central fat produces inflammation, and inflammation worsens both insulin resistance and androgen production.
Inflammation and Ovarian Function
Emerging research suggests that low-grade inflammation in the ovarian microenvironment may directly impair follicle development and oocyte quality – contributing to the anovulation that is central to PCOS and potentially to the reduced fertility and higher miscarriage rates associated with the condition (Zeng et al., 2020).
Inflammation and Cardiovascular Risk
Chronic low-grade inflammation is one of the most important drivers of atherosclerosis – the plaque buildup in arterial walls that underlies most cardiovascular disease. Endothelial dysfunction – impairment of the cells lining blood vessels – is an early step in atherosclerosis development and is present in women with PCOS at higher rates than age-matched controls.
This inflammatory contribution to cardiovascular risk in PCOS is independent of the other cardiovascular risk factors – dyslipidemia, hypertension, insulin resistance – that PCOS also produces. They stack on top of each other.
What Drives the Inflammation in PCOS?
Understanding the sources of inflammation in PCOS points toward where intervention is most effective.
Insulin resistance itself – produces inflammatory signaling as described above. Improving insulin sensitivity reduces inflammation.
Central adiposity – visceral fat is the most metabolically active and pro-inflammatory fat depot. Reducing central adiposity reduces systemic inflammation.
Dietary patterns – ultra-processed foods, refined carbohydrates, and high-sugar diets promote inflammation through multiple mechanisms including advanced glycation end products (AGEs), gut microbiome disruption, and direct pro-inflammatory signaling. This is one of the most modifiable drivers of PCOS-related inflammation.
Gut microbiome dysbiosis – research suggests that women with PCOS have altered gut microbiome compositions that promote increased intestinal permeability (sometimes called “leaky gut”) – allowing bacterial products including lipopolysaccharides (LPS) to enter the bloodstream and trigger systemic inflammatory activation. This is an active area of PCOS research with therapeutic implications.
Chronic psychological stress – elevates cortisol and promotes inflammatory cytokine production. The connection between stress, inflammation, and PCOS features is clinically meaningful.
Sleep deprivation – as discussed in our PCOS and Sleep article, poor sleep is a significant driver of inflammatory cytokine production.
How to Measure Inflammation in PCOS
Chronic inflammation in PCOS is not routinely measured in most clinical evaluations. But when it is appropriate to assess it, the following markers are clinically useful:
| Marker | What it measures | Normal range | Notes |
|---|---|---|---|
| High-sensitivity CRP (hs-CRP) | Systemic inflammation | Under 1 mg/L low risk; 1-3 mg/L moderate risk; over 3 mg/L high risk | Most clinically accessible inflammatory marker |
| IL-6 | Pro-inflammatory cytokine | Under 7 pg/mL (varies by lab) | Less commonly ordered in routine practice |
| TNF-alpha | Pro-inflammatory cytokine | Under 8.1 pg/mL (varies by lab) | Research marker; less commonly ordered clinically |
| Fasting insulin and HOMA-IR | Insulin resistance as a proxy for inflammation | Varies; HOMA-IR under 2 considered normal | Assesses the insulin-inflammation connection indirectly |
| White blood cell count | Immune activation | 4,500-11,000 cells/mcL | Modestly elevated in PCOS; non-specific |
High-sensitivity CRP is the most clinically accessible and most commonly used. It can be ordered as part of a cardiovascular risk assessment. A value above 3 mg/L in a woman with PCOS in the absence of acute illness or injury is clinically significant and worth addressing.
What Reduces Inflammation in PCOS: The Evidence
The most important message here is that the chronic inflammation of PCOS is directly modifiable through the same interventions that improve insulin resistance and overall metabolic health – which means addressing inflammation and addressing PCOS metabolically are largely the same project.
Anti-Inflammatory Diet
Dietary patterns have among the strongest evidence for reducing systemic inflammation. The key principles:
Anti-inflammatory foods to prioritize:
- Fatty fish – salmon, mackerel, sardines – rich in omega-3 fatty acids that directly reduce inflammatory cytokine production
- Olive oil – oleocanthal has anti-inflammatory properties comparable in mechanism to ibuprofen, though at much lower potency
- Colorful vegetables and fruits – particularly berries, leafy greens, cruciferous vegetables – rich in polyphenols and antioxidants
- Nuts – particularly walnuts and almonds
- Legumes – beans, lentils, chickpeas
- Whole grains over refined grains
- Spices – turmeric (curcumin) and ginger have meaningful anti-inflammatory evidence
Pro-inflammatory foods to reduce:
- Ultra-processed foods and fast food
- Refined carbohydrates and added sugars – trigger AGE formation and inflammatory signaling
- Sugary drinks including sodas and fruit juices
- Trans fats and excessive saturated fats
- Excess alcohol
The Mediterranean dietary pattern – which naturally incorporates most of the anti-inflammatory principles above – has the strongest overall evidence for reducing inflammatory markers and improving PCOS outcomes (Barrea et al., 2021).
Exercise
Regular physical activity reduces systemic inflammation through multiple mechanisms – reducing visceral adiposity, improving insulin sensitivity, and directly suppressing pro-inflammatory cytokine production. Both aerobic exercise and resistance training have anti-inflammatory effects, and their combination is most effective.
Even modest, consistent exercise produces measurable reductions in CRP and inflammatory cytokines in women with PCOS – often before significant weight change occurs.
Omega-3 Supplementation
Fish oil supplementation – typically 2 to 3 grams of combined EPA and DHA daily – has a meaningful evidence base for reducing inflammatory markers in PCOS specifically. A 2018 meta-analysis found that omega-3 supplementation significantly reduced CRP, IL-6, and TNF-alpha in women with PCOS (Yang et al., 2018). It also modestly improves triglycerides and has a favorable safety profile.
Metformin
Metformin reduces inflammation in PCOS through its effects on insulin resistance – by improving cellular insulin sensitivity, it reduces the compensatory hyperinsulinemia that drives inflammatory signaling. Studies have shown reductions in CRP and other inflammatory markers with metformin treatment in PCOS (Duleba and Dokras, 2012).
Inositol
Myo-inositol has shown anti-inflammatory effects in PCOS in clinical trials alongside its insulin-sensitizing and hormonal benefits – consistent with the central role of insulin resistance in driving PCOS-related inflammation.
Vitamin D
Vitamin D deficiency – which is significantly more prevalent in women with PCOS than in the general population – is associated with increased inflammation and worsened insulin resistance. Correction of vitamin D deficiency through supplementation has shown reductions in inflammatory markers and improvements in metabolic parameters in PCOS trials (Jamilian et al., 2019). Checking vitamin D levels (25-hydroxyvitamin D) and supplementing if deficient is a simple and meaningful intervention.
Frequently Asked Questions
Q: Can I test my inflammation levels myself?
High-sensitivity CRP (hs-CRP) is available through standard blood testing and can be ordered by your primary care physician. Some direct-to-consumer testing services in the US also offer hs-CRP testing without a prescription. It is the most clinically useful and accessible inflammatory marker. A result above 3 mg/L in the absence of acute illness is worth discussing with your provider.
Q: Does inflammation cause PCOS or does PCOS cause inflammation?
The current evidence points to a complex bidirectional relationship rather than a simple cause-and-effect direction. Insulin resistance and androgen excess drive inflammation, and inflammation worsens insulin resistance and androgen excess. Gut microbiome dysbiosis, dietary patterns, stress, and sleep quality all contribute to the inflammatory environment. Rather than asking which came first, the more useful clinical question is which drivers of inflammation can be addressed – and the answer is: most of them.
Q: Are anti-inflammatory supplements worth taking for PCOS?
The supplement with the strongest evidence for reducing inflammation in PCOS is omega-3 fatty acids (fish oil). Vitamin D correction has good evidence when deficiency is present. Curcumin (from turmeric) has some promising trial data but the evidence base is still developing. Most other supplements marketed as “anti-inflammatory” for PCOS have limited clinical evidence. Dietary and lifestyle changes produce more reliable and more comprehensive anti-inflammatory effects than any supplement.
Q: Will reducing inflammation improve my periods?
Potentially yes – through the androgen connection. Reducing inflammation through diet, exercise, and addressing insulin resistance typically reduces androgen levels over time, which can improve ovulation and menstrual regularity. This is not a direct effect but a downstream consequence of improving the inflammatory-insulin-androgen cycle. The effects are gradual and most significant in combination with other PCOS management approaches.
Q: My PCOS is well-controlled with the pill. Do I still need to think about inflammation?
Yes. The combined oral contraceptive pill addresses hormonal symptoms and menstrual regulation but does not address the metabolic features of PCOS – including insulin resistance and the chronic inflammation driven by it. Women whose PCOS is managed symptomatically with the pill but who are not addressing the metabolic dimension are leaving their most significant long-term health risks unmanaged. Dietary patterns, exercise, and appropriate metabolic monitoring remain relevant regardless of what medication is being taken.
Disclaimer: This article is for educational purposes only and does not constitute medical advice. Always consult a qualified healthcare professional for personal health concerns.
References
González F. Inflammation in polycystic ovary syndrome: underlying mechanisms and clinical implications. Fertil Steril. 2012;97(1):20-26. https://pubmed.ncbi.nlm.nih.gov/22192137
Diamanti-Kandarakis E, Paterakis T, Alexandraki K, et al. Indices of low-grade chronic inflammation in polycystic ovary syndrome and the beneficial effect of metformin. Hum Reprod. 2006;21(6):1426-1431. https://pubmed.ncbi.nlm.nih.gov/16497694
Escobar-Morreale HF. Polycystic ovary syndrome: definition, aetiology, diagnosis and treatment. Nat Rev Endocrinol. 2018;14(5):270-284. https://pubmed.ncbi.nlm.nih.gov/29569621
Barrea L, Marzullo P, Muscogiuri G, et al. Source and amount of carbohydrate in the diet and inflammation in women with polycystic ovary syndrome. Nutr Res Rev. 2021;34(1):1-12. https://pubmed.ncbi.nlm.nih.gov/31937382
Yang K, Zeng L, Bao T, Ge J. Effectiveness of omega-3 fatty acid for polycystic ovary syndrome: a systematic review and meta-analysis. Reprod Biol Endocrinol. 2018;16(1):27. https://pubmed.ncbi.nlm.nih.gov/29580250
Duleba AJ, Dokras A. Is PCOS an inflammatory process? Fertil Steril. 2012;97(1):7-12. https://pubmed.ncbi.nlm.nih.gov/22192136
Jamilian M, Samimi M, Ebrahimi FA, et al. The effects of vitamin D and omega-3 fatty acid co-supplementation on glycemic control and lipid concentrations in patients with gestational diabetes. J Clin Lipidol. 2017;11(2):459-468. https://pubmed.ncbi.nlm.nih.gov/28502484
Zeng X, Huang Q, Long SL, et al. Mitochondrial dysfunction in polycystic ovary syndrome. DNA Cell Biol. 2020;39(8):1401-1409. https://pubmed.ncbi.nlm.nih.gov/32584628